Corona vaccines are now travelling around the world. As refrigeration experts, German packaging manufacturers are helping to ensure that vaccines can be transported and distributed safely with their cool boxes.
One Study by DHL and McKinsey illustrates the challenges involved in transport: shipping ten billion vaccine units worldwide requires 15 million cool boxes are transported on around 200,000 pallets and in 15,000 freight flights.
Ultra Deep Freeze - ice cold is not cold enough
Due to the rapid development time, there is hardly any data on the chemical stability of such vaccines. To avoid risks, Biontech's active ingredient is therefore transported at a particularly low temperature of minus 70 degrees. Ultra Deep Freeze is what such temperature requirements are called in logistics. Moderna's vaccine has a slightly different protective mRNA coating than Biontech's, so a temperature of just minus 20 degrees is sufficient.
The right transport box for every temperature
Storopack offers several solutions that cover different temperature ranges, including transport down to minus 70 degrees. The boxes keep the temperature in the required cold range for a defined period of time during transport and also protect against damage. Cold packs or dry ice, for example, are used for cooling.
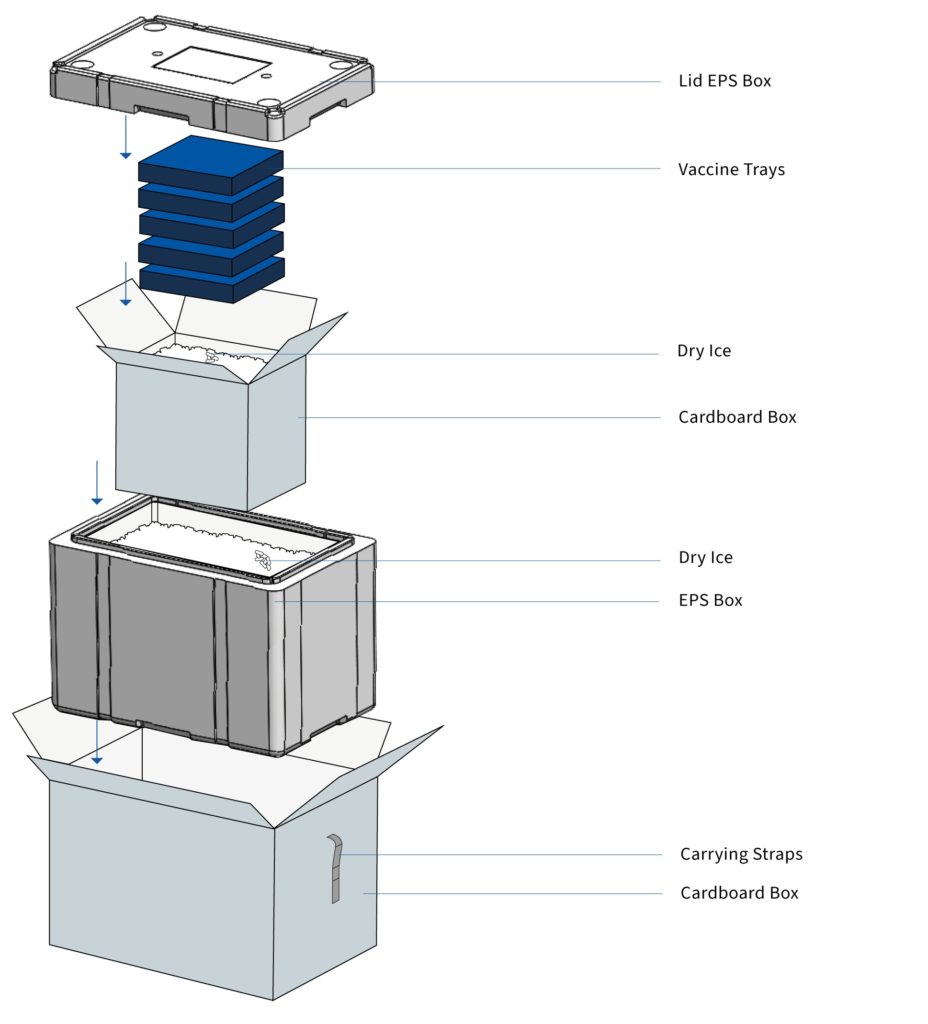
Of course, the shipping time also plays an important role. Depending on the requirements and the respective supply chain, Storopack's insulated boxes keep the required Temperature up to 12, 48 or 72 hours. This also enables transport within Europe through several countries if required. The boxes from Storopack are as a disposable or reusable solution and made of expanded polypropylene (EPP) or expanded polystyrene (EPS).
Corona vaccines require constant temperatures
Over a period of 250 hours, the O-Box H250 cold box from Ohlro temperature range between 2 and 8 degrees. The box with patented cooling system consists of a polystyrene box with combined cooling technology. In Vacuum units are located in the hollow chambers of all outer walls, which optimally prevent heat exchange with the outside world.

Under the Thermocon brand, the company develops and produces Schaumaplast Group a separate thermal packaging division for the pharmaceutical industry. When it became clear in the summer that the Biontech corona vaccine would have to be stored and transported at -70°C, the Schaumaplast engineers began to develop thermal boxes of various sizes. with dry ice as a cooling medium to qualify.
As a result, solutions with a product volume of one litre up to Insulated containers that hold a complete Euro pallet are ready. Even in summery outside temperatures, the -70°C is reliably maintained for more than five days in some cases. Thousands of vaccine doses have already been transported to their destination at a safe temperature in the polystyrene and neopor boxes.

As with all manufacturers of cool boxes for the worldwide transport of vaccines, the High cooling system requirementsThermocon refers to a special foam material, a special structure of the box and specially developed cooling liquids that release or buffer cold in doses.
The unusual grey appearance of the actual white EPS (airpop/Styropor) for the consumer results from the Addition of graphite, which further increases the insulation performance. Originally developed by BASF for the insulation of buildings, the material now makes an important contribution to the vaccine cold chain.

Also suitable for the transport of vaccines are the modular packaging from Feurer for the pharmaceutical and chemical industries. The packaging manufacturer has The world's first EPS hazardous goods packaging without outer carton developed. The airpop packaging, which consists of around 98 per cent air and two per cent plastic and is fully recyclable, is suitable for the worldwide transport of chilled materials in parcel shipments within 72 hours. The temperature range extends from plus 2 to plus 8 degrees and even down to minus 20 degrees.
SourceIK Industrievereinigung Kunststoffverpackungen e.V.