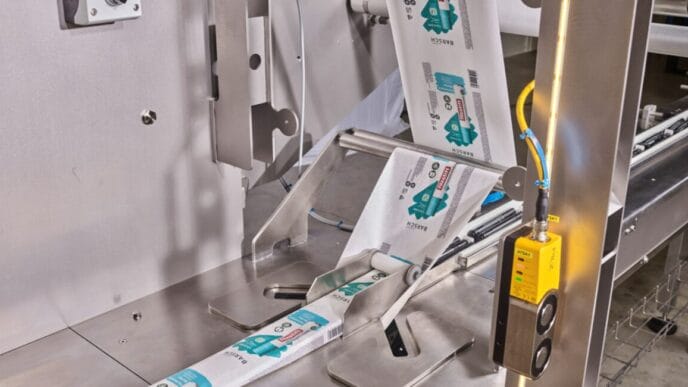
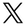The EU directive on combating counterfeit medicines poses challenges for the pharmaceutical industry because a much higher level of data management is required than ever before. According to the requirements, the packaging of most human medicines must have two new safety features: a unique identifier and protection against tampering.
Serialisation is performed on secondary packaging level required. The information for this must be provided as a 2D barcode and in human-readable form. The information provided must include a unique serial number, an expiry date, a batch number and a product code. There are currently differences in the classification of medicinal products. For example, a particular product may require a prescription in some Member States, while in others no prescription is required. The directive concerns only medical products that are prescribed. Therefore, depending on the destination country, an additional National reimbursement number required. This number is indicated accordingly by the manufacturers and the companies that repackage the medicines.

About a End-to-end testing system it must be ensured that the products are genuine. This system complements the risk-based verification by wholesalers. The final level of security is a further check of the product in the pharmacies. This ensures at the point of sale that the product in question is not a counterfeit. For products sold via the Internet, a new Online logo logo has been developed to recognise companies that comply with the applicable laws. By clicking on this logo, the user can ensure the authenticity of the company in question using a verified database. These new measures will come into force on 9 February 2019.
Labelling is an important component
The two most common technologies for serialised marking are laser and thermal inkjet (TIJ), as these technologies can be used to print high-resolution markings that meet the detailed requirements for 2D DataMatrix labelling fulfil. TIJ printers spray tiny droplets of ink onto the packaging as it passes by the cartridge or printer head. These ink droplets are ejected from a row (or several rows) of small nozzles using a fast-moving small heating element under the nozzles. These heating elements heat a small amount of ink, creating a small vapour bubble that propels the ink droplet onto the substrate. In contrast, laser coding systems use a focussed beam of light to mark or physically alter the top layer of the substrate. This beam of light is directed by two mirror galvanometers that align the laser beam on two planes.

Packaging substrates used
Cardboard and paper labels are often used, which frequently have a water-based coating to protect the packaging material. In the past, this would have been a problem for TIJ technology as water-based inks would not adhere to the surface of the packaging. Today, advances in ink technology have led to the availability of methyl ethyl ketone (MEK) and other light solvents, which have significantly increased the range of applications for TIJ technology. Substrates such as films, plastics and coated paper can now be processed using TIJ technology with the aid of MEK-based inks.
With laser systems can now be used to print on a wider range of materials, such as paper, plastic, metal and glass. Lasers can also be used to mark curved surfaces, such as vials and bottles. In the case of laser technology, two criteria must be taken into account when selecting a suitable substrate: Absorption of the laser light and creation of a print window with sufficient contrast for high-quality labelling. To achieve adequate labelling contrast, it is often necessary to use a print window with black ink (also known as „flood fill“) for the packaging. The laser burns off the top layer of dark ink, revealing the lighter-coloured substrate underneath - creating a negative image.

Focus on data management
The ability of labelling systems to interact with these technologies in different ways must be taken into account: With asynchronous communication, for example, the printer can send unsolicited information to the line control system. The function offers the dual advantage of active notification of printer events on the one hand and less data traffic on the network on the other, which in turn minimises the risk of errors. Faster notifications and a potentially higher throughput is possible. Remote communication protocols can also be used to exchange sensitive information and commands between the coding and marking system and the host data system. This is important when selecting a coding and marking system as it is critical that the printer has a proven, validated and secure protocol and command set specifically designed to manage sophisticated communications.

„Manufacturers will need to implement traceability technology to comply with the EU Falsified Medicines Directive and the associated delegated regulations,“ emphasises Arthur Smith, Global Marketing Manager at Videojet Technologies.
Buffer management aspect
Does the labelling system selected by the user support unbuffered labelling? In this case, variable data is received and each data set is printed individually. Or is the system suitable for buffered labelling, i.e. complete data packets are constantly sent, but then only printed once? These functions are particularly important in countries where serial numbers have to be purchased. Correct management gives manufacturers the opportunity to redeem unused numbers, which reduces expenditure.
Whichever technology is best for the particular requirements, one thing is clear from the legislation: labelling solutions for the EU Falsified Medicines Directive must offer robust data management, seamless integration into the production environment and high-resolution printing capabilities to provide the required information throughout the supply chain. By working with an experienced provider, manufacturers can ensure that the products they produce can be 100 per cent authenticated at every point in the supply chain. In this way, they can help to make the counterfeiting of medicines considerably more difficult in the future.
Videojet Technologies at ACHEMA 2018: Hall 3.0, Stand B95
[infotext icon]Falsified medicines and counterfeit drugsCounterfeit medicines are products that have been produced illegally and imitate real medicines. Basically, one can speak of Counterfeit products that purport to be the genuine medicinal product. The ingredients in these products may be of considerably lower quality than in the authorised genuine version. In addition, the active ingredient required for efficacy is present in lower doses. Generally, counterfeit packaging is used to conceal the origin of the product and the identity of the manufacturer.
Counterfeit medicines are often confused with counterfeit drugs and it is important to distinguish between the two. Counterfeit medicines are illegal, but are usually manufactured using patented technologies. This technology can be used to correctly manufacture medicines for which the manufacturer does not have the intellectual property rights or trademark rights. This turns the drugs into counterfeit medicines.
Counterfeit medicines, on the other hand, pose a higher risk to the public as they pretend to be genuine products. Unfortunately, the distinction between counterfeit products and falsified medicines is not made throughout the world as it is in the EU, and the terms are often used interchangeably.