Adents is a leading provider of unique product identification and traceability software that helps pharmaceutical companies and contract manufacturing organisations (CMOs) adapt to market changes and meet regulatory requirements for product traceability. The company was the first to advise on reliable Level 3 functionalities to centrally control serialisation at line level.
Adents offers a reliable, versatile and quick to implement Software suite for labelling and tracing product units, which enables the global tracking of each individual product anytime and anywhere. The serialisation solution is fully configurable and can be easily and flexibly adapted to technical and regulatory requirements. Adents was founded in 2007 by Christophe Devins and David Carpentier (CTO), who each have more than 25 years of experience in the development of platforms and systems for product identification and traceability.
Today, the company offers two solutions: Adents Seriza, The system consists of Adents Supervisor at site level and Adents Pilot, which can be used to implement serialisation and aggregation at line level. Serialisation solution for pharmaceutical manufacturers, which is recommended by Siemens for its pharmaceutical customers, among others. Adents Prodigi is a platform built on Microsoft Azure technology. Level 4/5 cloud solution, which was developed together with Microsoft. It enables secure data exchange and helps to fully exploit the potential of serialisation data.
„Our solutions for pharmaceutical packaging are primarily designed to ensure compliance with legal regulations and specifications. They also ensure seamless control throughout the entire supply chain,“ explains CEO and co-founder Christophe Devins.
Comprehensive Level 3 solution essential
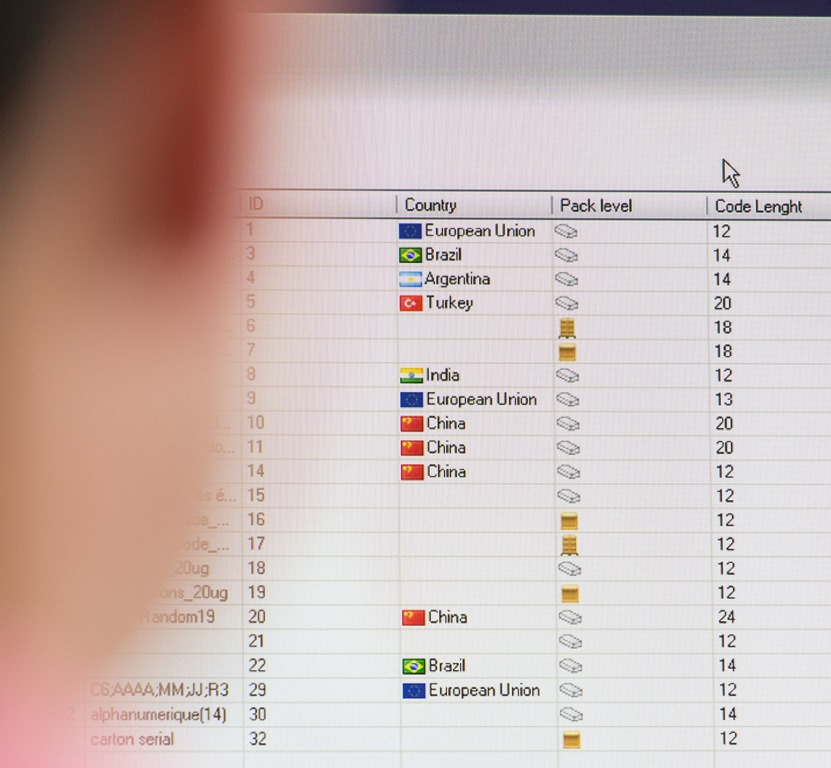
Serialisation solutions contain different levels: Level 1 generally includes line-level devices such as printers, scanners or cameras, Level 2 includes software for data control, serial number management and data aggregation for all Level 1 devices.
Level 3 refers to software systems at site level for transferring information from and to several Level 2 devices at each site. These systems manage the Production workflow for serialisation of the desired products: Master data such as information about customers, products and work orders. Reliable Level 3 systems are essential for the central control of serialisation data. A comprehensive Level 3 solution also offers Interface options to level 4 systems, to access and centrally manage work orders, delivery orders, product data, customer data, serial numbers (unless these are generated at Level 3), EPCIS events and XML files. Level 4 systems are often hosted outside the company firewall or in the cloud.
Thanks to Centralised configuration options With Adents Seriza, the level 3 system from Adents, pharmaceutical manufacturers can react quickly and precisely to changes without having to accept any loss of productivity. Adents Seriza also enables pharmaceutical companies and CMOs to manage customers, products and work orders. Adents Seriza generates serial numbers at site level in accordance with current regulatory requirements.

Change management support
A comprehensive Level 3 solution such as Adents Seriza offers versatile functions to support an effective Change management programme. Central configuration management prevents unauthorised or erroneous changes at line level by operators or supervisory staff.
For companies that follow current GMP, controlling and managing system changes is an enormous challenge. Cost point and a large The challenge is a challenge. For almost ten years, serialisation has been a buzzword that has unsettled pharmaceutical companies around the world. Varying requirements, legislative delays and a host of other factors are causing confusion and uncertainty around expectations for pharmaceutical manufacturers when it comes to securing their supply chains and providing data within their own jurisdictions and beyond. Thousands of sites and packaging lines will need to comply with legal and regulatory requirements as over 40 countries currently have serialisation regulations in place or soon to be. In the USA, for example, only the unique labelling of sales units with a data carrier must be guaranteed from November. By 2023, however, the Traceability of products throughout the supply chain be guaranteed. Although the European Directive 2011/62/EU harmonises the requirements for the European market, it leaves room for country-specific requirements. There are also draft laws in other countries and potential amendments.
How can companies successfully master such a transformation without disrupting operations? Serialisation solutions today must be able to meet the requirements of tomorrow and comply with ever new guidelines. The key to a future-proof serialisation solution is reliable functions at all technological levels that enable Implementation in several phases support. Software solutions should also be flexible and interoperable so that they can be implemented smoothly in existing packaging systems. With Adents Seriza, for example, plant managers can choose their hardware freely. Replacing individual components is also no problem in this way. Pharmaceutical companies and contract manufacturers must also ensure that they implement a centrally configurable Level 3 solution. This is the cornerstone of a successful serialisation programme.
Reduced validation effort without OEE losses
Implementing new industrial processes at different production sites and lines is a difficult endeavour that can require extensive change control. With Adents Seriza, all necessary processes (serialisation, aggregation, etc.) can be configured centrally. Work orders are managed at Level 3 level and transferred to Level 2 systems using a one-to-many distribution model. The centralised Level 3 configuration provides a higher level of governance and reliability in modern, dynamic environments. Only when Parameters configured centrally can be made quickly and smoothly. Centralised configuration at level 3 avoids unnecessary changes and adjustments to the code. New information is automatically passed on to the individual lines, i.e. there is no need to reconfigure and validate each line individually.
Any upgrades can also be carried out quickly. Adents Seriza ensures that the system is always up to date with the latest configuration changes without any operational downtime. No intervention is required at line level, so the process runs completely autonomously and human error is eliminated.
With serialisation solutions that are limited to line level, the testing and validation process is often carried out by Documentation requirements complicated. A practical Level 3 solution offers standardised applications at line level with an architecture that ensures low impact, easy access and documentation and low risk in terms of validation. This becomes all the more important the more lines a production facility has to equip. Standardised applications at line level with central configuration at site level make Level 3 solutions such as Adents Seriza scalable and quick to install, as the installation on a pilot line can be easily transferred to other lines and does not have to be validated each time.
The centralised configuration of work orders also facilitates transitions and production changes and helps to avoid production downtimes. By automatically transferring centrally parameterised process information to the line in advance, the next work order can be started immediately after completion without having to re-parameterise and test the line.

Practical example CMO
The task: One of the five largest CMOs in Europe with a dozen production sites decided to implement a serialisation solution on seven packaging lines in four different plants to meet the pharmaceutical serialisation requirements set by the USA, Brazil, South Korea and China. The customer wanted to implement serialisation of marketable units for these four markets (especially South Korea and China) in the short term and be able to expand its plant to meet the serialisation requirements of ten other European countries without further development effort. The CMO plans to equip a total of 80 lines to meet the increasing demand for serialisation due to expiring deadlines in the US and Europe.
Manual lines process 40 items/minute, semi-automatic lines 300 items/minute. In order to continue using the existing equipment and to be prepared for future implementations and upgrades, the company selected Adents Seriza and implemented the solution at four different sites in France
The result: Ten different packaging formats can now be processed on a single line. Thanks to the high reliability of Adents Seriza (even at high speeds) and the easy management of product transitions, the customer is experiencing no loss of productivity on lines that now not only pack but also serialise products. In the first year after implementation, ten million products were serialised and aggregated according to four different regulations. The contract manufacturer now provides serialised production for at least four pharmaceutical companies and has the capacity to meet even greater demand and customer requirements.
[infotext icon]Adents, software specialist for the serialisation and traceability of pharmaceuticals, is an internationally operating company with offices in Europe and the USA and a global network of solution partners.